Back in my undergraduate days I remember finding the CRC Handbook of Laboratory Safety. One photograph really stuck with me. Years later I decided to replicate it and use the image in chemical safety training.

The picture above shows what happens when a solution of soluble proteins in water is subjected to a large excursion in pH in both directions- both highly acidic and highly basic (caustic). The take home lesson is intended to be “wear your damned safety glasses or face shield”. The obvious comparison is between egg white and your corneas. Both have transparency and proteins.
In reality, there is no comparison between the composition between an egg white and a cornea. The human cornea is far, far more complex in composition and structure. Eggs are widely available and cheap. While we see many human corneas every day, they are attached to living people who would, no doubt, put up a tussle with anyone seeking to abscond with one. For a demo, egg white will have to do.
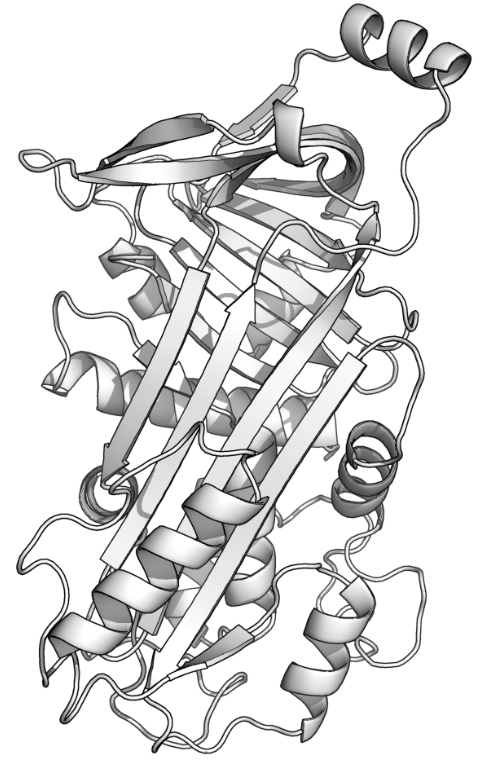
The major protein in egg white is the globular phosphoglycoprotein ovalbumin at 54 % abundance. According to Google it is “A major storage protein with phosphorylation properties.” Ovalbumin and human serum albumin share a name but little else. Ovalbumin serves as a storage protein source for developing chicks. Human serum albumin serves to maintain balance in the osmotic pressure of blood and to transport substances in the blood stream.
The egg white image is really about the effects of corrosives on protein. Ovalbumin exposed to strong acid will rearrange its globular structure in a way that renders it insoluble and causes agglomeration. Thus the opaque appearance. Trainees look at the image and, knowing they themselves are made of protein, can silently draw their own conclusions about the risk of getting a corrosive in their eyes.
