I’ve come around on this business of the atom being almost entirely empty space. This is an established bit of folklore in intro chemistry and physics. It dates back to experiments by Hans Geiger and Ernest Marsden under Ernest Rutherford, showing how alpha particles could sail through thin gold foil and infrequently, an alpha particle would impact something hard and scatter. The striking thing about the experimental results was just how infrequent the scattering was. The conclusion eventually drawn was that the atoms in the gold were mostly empty space.
But what if that space wasn’t quite empty? What if that space was a beehive of electrons at maybe half light-speed and mutually repelled by one another yet attracted to the nucleus. Each electron is a single point negative charge. The nucleus has a diameter 100,000 times smaller with equal but opposite charge. The strong positive nuclear charge field holds the electrons tightly but only to the to the point where electron-electron repulsion is balanced in atoms with more than two electrons.
The electron is a point charge manifestation of the electromagnetic force, but with mass and angular momentum. It is a perturbation in the electric field. It doesn’t fly like a ball, it exists in the manner of a wave of chance. It has none of what humans think of as material substance, rather it is purely a quantum mechanical manifestation. It is shaped by 3-dimensional standing waves of probability density surrounding the nucleus. This probability density is defined by a spherical harmonic wave series. We chemists know this harmonic series as s, p, d and f “orbitals”. Electron probability density extends from the nucleus to the outer orbitals of the atom with s, p, d, and f orbitals occupying space defined by their unique wave equations.
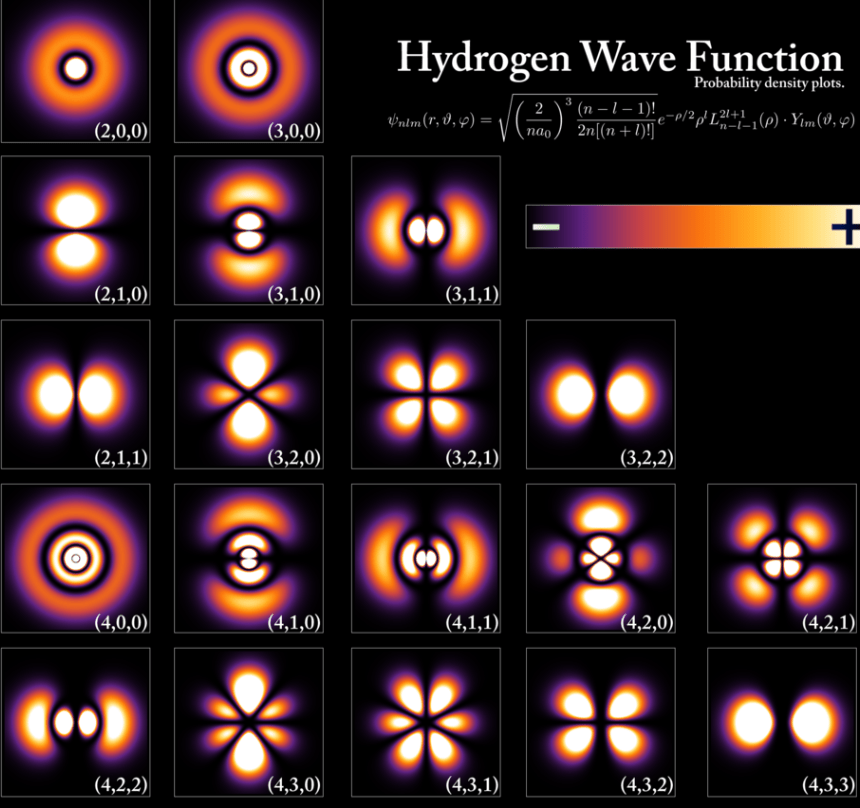
As a reminder, the shape of an orbital itself defines a region of space where an electron of a certain energy is most likely to be found. It is not necessary to be able to calculate the position of the electron moment to moment to understand its properties. Heisenberg’s Uncertainty Principle does not allow for high precision determination of both position and momentum simultaneously, so this is where the universe tells us that ‘ya can’t have everything’. However, energy levels and transitions between them can be measured precisely. Exact position of an electron is not necessary. Besides, the 3-body problem shows up very early in the periodic table and spoils the fun anyway.
The edges of orbitals are not sharp but rather feather off into space and are pragmatically defined by a reasonable certainty as encircling an overall 95 % probability density.
What about the ’empty space’ view of the atom? As previously surmised, the filled concentrically overlapping occupied orbitals of an atom define a region of electron probability density that is not ever empty except for the hydrogen cation, H+.
Recall that the mass of the electron is small, about (1/1800)th that of the proton mass. This says that the space between outer edge of the atom and the nucleus is occupied by the electrons which are in constant motion constrained only by the individual 3-dimensional orbitals.
This forces us the think more clearly about what constitutes ’empty space’ of the atom. That empty space is filled with a diffuse, low mass density swarm of negative charges. Only orbital nodes have zero electron density, but all orbitals have some probability density throughout the interior of the atom.
Perhaps better way to describe the space between electron and nucleus is to simply mention the dimensions of the atom and its nucleus in meters as an example.
And for the Rutherford gold foil experiment, the diffuse electron density around the nucleus would pose little resistance to an alpha particle with its larger momentum passing through, giving the illusion of empty space.
A gold foil of larger thickness will easily block all alpha particles. Alpha’s are stopped by losing their energy to ion formation when passing through matter.
