C&EN has a web page devoted to a linked bibliography of safety-related letters to the editor. It is worth having a look at. It is good to have a healthy interest in energetic reactions and incompatible substances.
Category Archives: Chemistry
A critique on scale-up suitability
In my quest to stimulate bench chemists to think like industrialists, I like to bring examples of chemistry from the literature to highlight a point I’m trying to make. The literature is full of transformations and research that serve as positive and negative examples of good scale-up thinking.
There are examples, however, that are less than choice in terms of green processing or good scale-up thinking. As I have said previously, green chemistry and good scale-up principles may not be equivalent concepts, but they can and often do run in parallel.
An interesting transformation is featured in the recent article entitled Efficient 1,2-Addition of Aryl and Alkenylboronic Acids to Aldehydes Catalyzed by the Palladium/Thioether-Imidazolinium Chloride System, by Kuriyama, Shimazawa, and Shirai, J. Org. Chem., 2008, 73, 1597-1600. [My apologies to the authors for their unanticipated role in this analysis.]
In this article a bond forming reaction between 1.5 eq of a boronic acid and 1.0 eq of an aldehyde is described affording a secondary alcohol. The transformation is catalyzed by 0.5 % Palladium allyl chloride dimer with 1 % of a custom imidazole carbene precursor in the presence of 2 eq CsF as base. The reaction mixture is heated to 80 C in dioxane and the chemistry is reported to be over in ca 20 minutes.
I am somewhat reluctant to be critical of chemistry that is done catalytically and is high yielding. But this transformation, solid science though it may be, would be difficult to justify taking to scale-up without an examination of alternative schemes. Let me explain my thinking.
First, on the basis of atom efficiency alone, this process requires that a lot of different elements find their way into the pot. The tally is C, H, N, O, Cl, B, Pd, Cs, F, and S to just make a C-C bond to produce a benzyl alcohol. A scale-up chemist would have to ask, why not use a Grignard and the aldehyde? Granted, there may be incompatible functional groups on either Ar1 or Ar2 that would not tolerate a Grignard reagent. However, it is worth pointing out that the conventional way of making boronic acids is by addition of a boronic ester or fluoride to RMgX or RLi followed by hydrolysis. Compatibility is an issue there as well.
One might object that many of the diverse atoms used in the reaction are at a catalytic level and as such may not constitute a major cost or environmental insult. True enough for the user of the process. But the metal complex must be manufactured somewhere at a larger scale for distribution. Pd mining and beneficiation requires energy inputs and generates wastes. The same idea applies to the imidazolinium salt.
The reaction does seem to require 1.5 equivalents of boronic acid and 2 equivalents of cesium fluoride. Boronic acids are specialty synthetic intermediates whose manufacture generates its own waste stream. Furthermore, boronic acids can be on the expensive side. The use of a boronic acid as a latent nucleophile for a straightforward addition to an aldehyde seems somewhat extravagant.
Cesium fluoride residues (2 equivalents) will find their way into the aqueous waste stream and possibly to an incinerator where the solids may end up in roadway pavement or a landfill. While fluoride is an efficient base in this case, common sense suggests that carbonate may have a more benign fate in the environment owing to the fact that it decomposes to water and CO2. Unfortunately, the best yields are with cesium as cation.
Chemists seeking to apply this kind of coupling chemistry would be well advised to be extra careful in their IP diligence. The use of metal catalyzed coupling reactions may already be patented or applications may be pending for patents. The same comment applies to the use of imidazolinium carbenes. Industrial chemists would be well advised to look deeply into the carbene species for process and composition of matter claims. Ever since the Bayh-Dole Act, university patents have been popping up like dandelions.
I do not want to be too critical of this chemistry. It is an interesting transformation and certainly may be of use for some kind of product. But for scale-up, at first pass it seems too far from earth, air, fire, and water. I would say that for maximum profit, this process is more of a Plan B or Plan C scheme.
How to pass organic chemistry
WordPress shows the blogger what search terms lead the searcher to your blog. One of the searches that lead a reader to this blog was “How to pass organic chemistry”. Here is my answer-
Don’t get behind. Study study study study study study study study study study study study do the problems study study study study study try to enjoy it a little study study study study study study study study study study study study study study study study study study study do the problems study study study study study play racketball study study study study study study study study read the chapters 3 times study study study study study study study study study study study study study study study understand exactly what the problem asks study study study study study try to enjoy it a little study study study study study study study study study study study study study study study study study study study do the problems over again study study study study study try to enjoy it a little study study study study study study study study go back and scan an earlier chapter study study study study study study study study study study study study study study study study study study study take a few breaks study study study study study study do the problems study study study study study form a study group study study study study study study buy the solutions manual study study study study study study study study study study study study study study study study study study study study study study study study study study do the problems again study study study study study form a study group study study study study study study study study study study study study study study study study study study study do mechanisms on a blackboard study study study study study go dancing study study study study study study study study read the chapters 3 times study study study study study study study study study study study study study study study buy a model kit study study study study study try to enjoy it a little study study study study study study study study study study study study study study study study study study study find a girlfriend study study study study study try to enjoy it a little study study study study study study study study learn to draw structures using perspective study study study study study study study study study study study study study study study study study study study study study study study study study study study take a pottery class study study study study study pay attention study study study study study study study study learn the mechanisms study study study study study study study study study study study study study study study study study study study study study study study study study study do the problems study study study study study try to enjoy it a little study study study study study study study study study study study study study study study study study study study look up the structure of a medicine study study study study study try to enjoy it a little study study study study study study study study read the chapters 3 times study study study study study study study study study study study study study study study do the problems study study study study study go have tacos at 1 am study study study study study study study study study study study study study study study study study study study try to draw 3-D renderings of the structures study study study study study try to enjoy it a little study study study study study study study study read the chapters 3 times study study study study study study study study study study study study study study study study study study study take a few breaks study study study study study study do the problems study study study study study attend a study group study study study study study study learn the mechanisms study study study study study study study study study study study study study study study study study study study study study study study study study study visit the prof during office hours study study study study study try to enjoy it a little study study study study study study study study study study study study study study study study study study study go have beer & pizza study study study study study recopy your notes study study study study study study study study read the chapters 3 times study study study study study study study study study study study study study study study do the problems study study study study study try to enjoy it a little study study study study study study study study study study study study study study study study study study study go out on a date study study study study study push electrons study study study study study study study study summarize each chapter study study study study study study study study study study study study study study study study study study study study study study study study study study study do extra problems study study study study study try to enjoy it a little study study study study study study study study practice drawing structures study study study study study study study study study study study study study study. Get some sleep.
1950’s Chemistry
I recently spent some time listening to an acquaintance talk about his days as a student at MIT and as a grad student at Harvard in the early 1950’s. He had Geoff Wilkinson for inorganic chemistry at MIT as an undergrad and later did his PhD with Wilkinson at Harvard. Curiously, Wilkinson did radiochemistry in the Manhattan Project prior to joining academia. His radiochemistry experience compelled him to work fast and in test tubes, according to my friend.
My friend’s lab mate in Wilkinson’s group was Al Cotton. They started grad school together ca 1952 or so. This was shortly after the sandwich structure of ferrocene was proposed by Wilkinson’s fellow Harvard prof R. B. Woodward. Woodwards basis for this structure was on symmetry and a single IR stretch absorption. Spectroscopically, the original sigma bonding model didn’t fit the data. Just prior to this, Wilkinson had begun work on a variety of organometallic Cp compounds. As the story goes, when Woodward expressed interest in making more Cp compounds, Wilkinson went to his office and “had words” with Woodward. Afterwards, Woodward moved on to other things.
My friend laughingly recalls the time he was chewed out by his P-Chem prof, the great George Kistiakowski and earlier, by Arthur Cope at MIT. He recalls being summoned to Cope’s office. Cope was wearing pink slacks which contrasted with his red hair. He was displeased about the impertinent back channel invitation my friend pitched to Linus Pauling to speak to the chemistry club. (I haven’t verified the color of Cope’s hair)
My friend recalls having E. J. Corey as a lab assistant while in an undergraduate lab at MIT. He joked that he saw Corey once at the beginning of the term and once at the end. My PhD advisor, Al Meyers, did his post doc with Corey some years later. Small world.
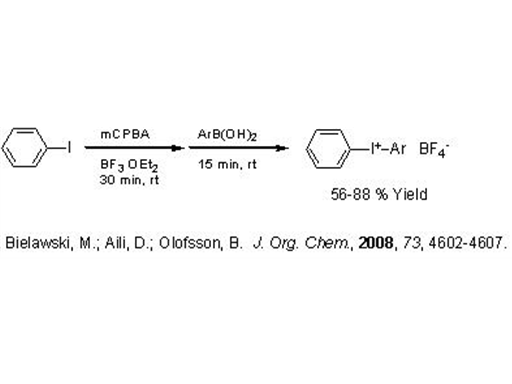
Preparation of Iodonium Tetrafluoroborates
An interesting bit of chemistry was published by Berit Olofsson at Stockholm University in a recent JOC. The Olofsson lab has previously produced a method for the one-pot preparation of diaryliodonium triflates. This latest work provides diaryliodonium tetrafluoroborates (JOC, 2008, 73, 4602-4607).
The preparation of I(III) compounds usually starts with an Ar-I compound undergoing oxidation followed by an electrophilic addition/substitution to another arene. Regioselectivity is obtained by choosing a donor with a leaving group such as a boronic acid, stannane, or silane.
What is clever about this process is the fact that a BF4 salt is directly produced. Two equivalents of boron trifluoride etherate are used in the reaction which evidently results in some kind of disproportionation producing the BF4 counter-anion.
It is known that the reactivity of iodonium compounds is somewhat sensitive to the coordinating ability of the counter-anion, so BF4 is less undesirable than other choices (like chloride). Solubility is greatly influenced by the choice of counter-anion as well. This is particularly true in photo-initiator applications where the choice of carrier fluid may be limited.
Meat and Cheese Explosives
So, I’m blundering through the literature on a snipe hunt when I run into this ICI patent- US 5,456,729. In the description, they teach a method of preparing an explosive composition using “lactic casein”. Having been in the dairy business long ago, and specifically having worked in a cottage cheese plant, I recognized this component as … cheese. Well, mostly. Example 5 discloses a composition comprising 25 % ammonium nitrate and 3 % lactic casein.
Unless you have lactose intolerance, cheese is not ordinarily an explosive. In the patent, the lactic casein is one of many examples of a foam stabilizer. Other stabilizers include animal and fish proteins as well as collagens. A collection of other chemical additives rounds off the list.
If they had specified gluten, they could have claimed the use of a pastrami and cheese on rye sandwich as stabilizer feedstock for their explosive composition.
Structural diversity of organic chemistry
The recent issue of Journal of Organic Chemistry, (JOC, 2008, 73(12)) has a few articles that are particularly interesting.
The article by Lipkus, et al., entitled Structural Diversity of Organic Chemistry. A Scaffold Analysis of the CAS Registry, JOC, 2008,73, 4443-4451, is a particularly ambitious bit of work that only CAS could do. This article describes a scaffold survey of more than 24 million organic compounds in the CAS Registry.
The data set was limited to carbon-based structures containing the heteroatoms H, B, Si, N, P, As, O, S, Se, Te, and the halogens. Moreover, the work was further limited to framework structures containing rings or linked rings. Acyclic compounds were not included owing to the inapplicability of the framework definition in the search algorithm. Multicomponent substances and polymers are ignored as well.
Lipkus and coworkers found that half of the graph frameworks analyzed are described by only 143 framework shapes. The remaining half are described by 836,565 graphs.
One of the key conclusions is quoted here-
“It is not surprising that some frameworks occur much more frequently than others. However, the extreme unevenness in the way frameworks are distributed among organic compounds is somewhat surprising. This is particularly true at the graph level, where it is found that only 143 framework shapes can describe half of the compounds. The fact that both graph and hetero frameworks have very topheavy distributions tells us that the exploration of organic chemistry space has tended to concentrate on relatively small numbers of structural motifs.”
Lipkus concludes that cost minimization is one of the drivers of this “… shaping the known universe of organic chemistry.” He comes to this conclusion due to the presence of a power law which describes this distribution. The power law he refers to is a linear log-log relationship that is indicative of what they refer to as the “rich-get-richer process”.
If I understand this correctly, a relatively small number of easily made or commercially available early precursors are comprised of ring graphs that, by virtue of modification, propagate into more complex analogs that retain the original graph. This has the effect of multiplying the frequency of a given graph.
The cost minimization aspect comes from the benefits of familiar chemistry and the commercial availability of a fairly limited set of ring graphs. Adding more rings will usually mean adding more molecular weight and adding problematic synthesis and separation issues.
The authors conclude that the lopsided distribution of organic compounds toward only 143 graphs comprises a bottleneck in drug discovery. They further suggest that more exploration in other areas of chemistry space may be worthwhile.
My dinner with a meteorite
Last night I found myself sitting at a restaurant with astronomers for the occasion of viewing a meteorite. Customarily, a few observatory folk have dinner with the speaker and then we go to the observatory for a public star night. While waiting for our entrees we passed the object carefully amongst ourselves, cherishing a few moments of close contact with this rare object.
Astronomers seem to be prone to public displays of humility. I would estimate that the humility quotient was near 0.8 (8 out of 10 Sagans- the Sagan is the international unit of humility). It is generally agreed that the Buddha achieved a Sagan quotient of unity. Okay, I’m kidding.
The curious 936 gram achondrite is from the recent Berthoud, Colorado, fall. Meteor enthusiasts refer to the arrival of a meteorite as a “fall”. This is one of only 5 witnessed falls in Colorado. A section of the meteor has been cut off and has been the subject of investigation at the University of Arizona.
Based on the composition of the object (olivine, plagioclase, ilmenite, chromite) and based on the reflectance spectra of various asteroids, the Berthoud meteorite is thought to be a fragment of the asteroid Vesta. Imagery of Vesta suggests that a portion of this object may have been shattered by an impact in the past.
The family whose property the stone landed on are somewhat bewildered by the event. They have been the subject of much unwanted attention, so the object is kept secure at an unknown location. In October of 2004, in the early afternoon several family members were standing outside their home when they heard a whistling sound and thump. Following the direction of the sound, they found the impact site less than 100 feet from where they were standing and in a spot where one member had just walked through. Only a small part of the object protruded upward through the disturbed topsoil.
Reportedly, it was cool to the touch immediately after arrival. This is counter-intuitive given the fiery appearance of most meteors. However, the object was quite cold prior to entry into the atmosphere and the rapid transit through the air didn’t allow for heat saturation. And, ablation carries away much of the friction energy.
The low iron object has a dark fusion crust over a grey, mottled composition. Other than the crust, it is not that unusual in its appearance.
Verbund Manufacturing
German manufacturing culture does many things very well, but a few things particularly stand out. One of these items pertains to the concept of verbund manufacturing. Verbund simply means “integrated” or “linked”. Verbund manufacturing sites are clusters of manufacturing units that take advantage of proximity. Clustering can offer certain logistic and energy advantages if done intelligently.
A cluster of manufacturing sites can operate and share a co-generation plant for the distribution of steam, waste heat, and electricity. Large capital items like steam plants can be shared so funds can be plowed into larger scale for better economy. Rail operations and other transportation resources can be shared as well. Clustering also provides for the possibility of vertically integrated manufacturing on site and a reduction in transportation costs.
Clustered manufacturing may also have the effect of concentrating the supply of skilled workers for the labor pool. A manufacturing nexus can attract community colleges and other vocational opportunities for the next generation of employees.
The USA has many manufacturing sites where similar industries congregate. Look at the Gulf coast with all of the refinery locations. But the extent to which there are synergistic interactions between companies is unclear.
In the US, corporations tend to behave as the Republic of Exxon or the Republic of the Union Pacific. This kind of a fragmented confederation of corporate states is becoming obsolete as we go up against nationalized business entities that control key resources and trade. The key to future vitality is greater efficiency with resources. Synergistic cooperation is one model that is available. But to do this requires trust and the desire to cooperate for mutual benefit. Competition begets gamesmanship and posturing which works against the verbund model for US businesses.
US corporations have much to learn from this business model.
“60 Minutes” and Dust Explosions
Sunday evening on 60 Minutes on CBS there was a segment on dust explosions. For the most part, it was an expose on the failings of OSHA. It is hard to avoid the conclusion that OSHA is lead by a bunch of dullards who are under the enchantment of an administration reluctant to impose new regulations on industry.
The thrust of the program was that OSHA is completely unable to recognize incipient dust hazards on their site inspections, partly due to a lack of training and partly due to a slack-jawed lack of direction. It wasn’t pretty.
As a dramatic backdrop, numerous instances of major plant explosions were trotted out for all to see. The message is that plants keep blowing up from dust explosions, but OSHA isn’t holding companies to higher standards- because there aren’t any. The Secretary cited OSHA’s housekeeping requirement as broad enough to cover the dust explosion scenario. It was less than convincing.
I couldn’t help but notice that the subtext was that there can only be safety if more regulations were written. I didn’t see any company officials grilled in the same manner that the Secretary was grilled.
In fairness to OSHA, someone needed to clarify just what that agency is free to do in regard to rule making and what must be done by the Congress. I know there are smart people in OSHA, but being federal employees, there is little incentive to champion new regulations. Between institutional inertia, lobbyists, and an antagonistic executive branch, who wants to charge ahead of the parade on new rules?