I’m saddened to learn of Emeritus Professor of Chemistry Michael P. Doyle’s passing. Mike had retired recently from the University of Texas San Antonio. Those who knew Mike knew that he was a dynamo of research productivity in undergraduate institutions and later in a PhD granting institution. In his career, Mike had 439 or more papers published (Google) in prominent publications. One of his secrets to productive research in 4-year institutions was the use of multiple postdocs who guided his undergraduate researchers in their work. Together they performed synthetic organic and organometallic chemistry and produced a rich tapestry of rhodium catalyzed carbene transformations.
Like research faculty everywhere, Mike’s success in grant writing was key to keeping up a productive lab and attracting excellent post-doctoral chemists. His publications and fascinating catalysis chemistry as well as his participation in the many national institutions of chemistry kept him near the leading edge of his field.
I was a postdoc in the Doyle lab from 1990 to 1992. My interest was in developing a career path similar to Mike’s and his lab was the place to be. During my time with him at Trinity University, Mike’s interest was in asymmetric catalysis and my part in that was asymmetric C-H insertion into secondary carbons with diazoacetates and a chiral dirhodium paddle wheel complex. We used chiral Pirkle-type GC columns to determine the enantiomeric excesses (% ee) of many of our products. The undergrads caught on to this right away and could run a reaction and get GC and NMR results right away on their own.
During my time with Mike at Trinity, the university had a visit and a talk before the whole university community by Margaret ‘Maggie’ Thatcher, former Prime Minister of Great Britain. Of interest is the fact that Maggie was a chemist by training. That day she toured our labs to see what Mike’s research was all about.
He held positions as a Professor at institutions including Hope College, University of Arizona, Trinity University, and the University of Maryland. He has also been a visiting professor at the University of Iowa (Google).
During his career Mike participated in many professional organizations and won numerous awards. He served in the executive committee of the ACS Division of Organic Chemistry for 23 years in many capacities as member, councilor as well as its chair. He served as chair of the Executive Board of Chemical & Engineering News (C&EN). He was a founder and first president of the Council for Undergraduate Research and first chairman of National Conferences on Undergraduate Research. He was also president of Research Corporation for Science Advancement.
Mike was immortalized with one of the highest recognitions of all, a named chemical reaction: the Doyle–Kirmse Reaction.
Fellow of the American Chemical Society
2013 – Fellow, National Academy of Inventors
2010 – Fellow of the American Association for the Advancement of Science (AAAS)
2009 – Fellow of the American Chemical Society
2003 – Member of the National Academy of Medicine (NAM)
1994 – Fellow of the American Association for the Advancement of Science (AAAS)
2009- Fellow of The Royal Society of Chemistry
2006- Arthur C. Cope Senior Scholar Award
2002- Pimentel Award in Chemical Education in 2002 from ACS
1998- Gassman Distinguished Service Award from the Division of Organic Chemistry
A list of citations in Google Scholar can be found here.
Probably the most prominent of the Doyle catalysts is Rh2(5S-MEPY)4, or dirhodium(II) core surrounded by four bridging chiral methyl 2-pyrrolidinone-4(S/R)-carboxylate (MEPY) ligands. In the lab it was just known as “MEPY”. It was a real workhorse, useful in several types of chemistries including cyclopropanation, cyclopropenation, Lewis acid catalysis and C-H insertion.
MEPY catalyst was prepared from commercial dirhodium tetraacetate and ligand and performed in a Soxhlet extraction setup with sodium carbonate to remove the acetic acid produced. It could be cleaned up by column chromatography.
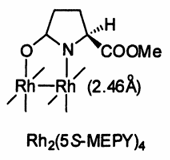
The Doyle Rh2(5S-MEPY)4 catalyst showing the dirhodium core with 1 of 4 chiral, enantiomerically pure, methyl 2-pyrrolidinone-4-(S/R)-carboxylate ligands. The above structure was captured with acetonitrile ligands in the two axial positions.
Mike’s gift for academic research was built on his boundless energy and friendly nature. He loved working with undergraduate chemistry majors and nearly all got their work published in prestigious journals. We postdocs participated his research model and methods for our future academic careers.
I had the opportunity to visit with Mike at his home in San Antonio 2 years ago on the occasion of his retirement from UTSA. He was in good spirits and spent time gabbing with each of us. Glad I went.
Editorial note: I fixed nomenclature of MEPY. Excuse me.


I’m sorry to read of your loss. It is hard to say that goodbye to a mentor.
Thanks Morris. I hope everything is going well for you. I’ll be retiring May 1st. The local newspaper is trying me out as a contributor. I decided that I wasn’t getting enough hate mail …